CROs

What elements of Informed Consent must we include when pre-screening?
Blog Posts
Clinical Endpoints
The Role of Expert Committees: An Interview with Dr. Seltzer
Videos
Ethics in Clinical Research
Does the IRB need to review news stories?
Blog Posts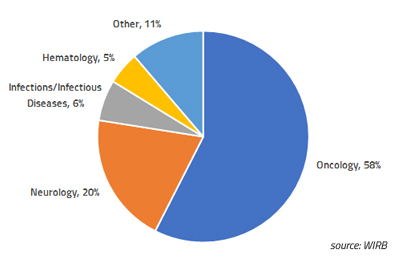
Single-Patient Expanded Access: WIRB experience in 2018
Blog Posts
Limited IRB Review: Are You Prepared for January 21st?
Blog Posts
Clinical Endpoints
Error Rates and Data Integrity: eCOA versus Paper Administration of the PANSS
Research
Site Efficiency
IRBs can make the most of central IRB partnerships
Articles
Clinical Trial Safety
Infectious Disease Challenge Studies: Ethical Issues in Causing Disease for Medical Knowledge
Whitepapers
WIRB-Copernicus Group Acquires KMR Group and Metrics Champion Consortium (MCC)
News
FDA & ICH